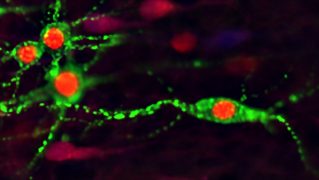
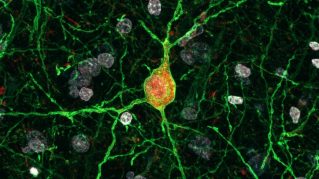
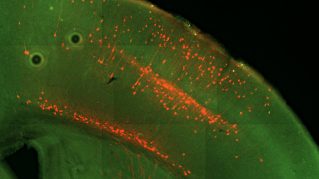
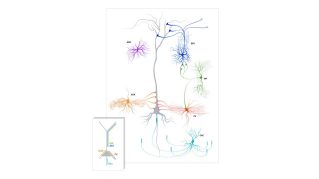
Cortical Interneurons
The originally funded CGSB joint initiative between the Desplan and Fishell laboratories aimed to determine the unique genomic expression profiles of neuronal subtypes in Drosophila and Mouse. Significant progress was made by examining the expression profiles across neuronal populations within these two species and then through the use of single cell RNA-seq approaches to examine gene expression within individual neuronal populations. To address how these unique genetic profiles are generated, we will examine the developmental progression of gene expression in progenitors from their inception to their integration into brain circuits. Our studies in the mammalian forebrain have already revealed the surprising insight that early progenitors of the various classes of interneurons initially share a large proportion of their transcriptomes while the differences observed in the mature interneurons evolve only gradually across development. Therefore, the diversity of mature interneurons appears to result from expression of a relatively small number of “cardinal determinant” genes initiated during development. To decode this diversity we will identify candidate genes using an innovative epigenetic screening method, as well as testing the role of cardinal determinant genes using CRISPR technology. Studying the role of specific subtypes of neurons in development and disease is currently limited to the use of transgenic animals. To increase the flexibility of these approaches and extend them to non-genetically amenable species (i.e. rats and non-human primates), we established a viral core that produces both recombinant adeno-associated virus (AAV) and the recombinant rabies virus (RV). Each of these viral vectors provide significant utility for the study and manipulation of mammalian neural circuits. While AAVs provide a vector where expression can be directed with a growing array of available small CIS-acting elements that provide celltype specificity, Rabies viruses can be used to tracer and manipulate monosynaptic circuits. Together the Fishell laboratory at NYUAD provides an essential resource for complementing the labs study of cell types in the mammalian forebrain and allows the CGSB program to provide reagents that allow their efforts to be at the forefront of neuroscience. The pipeline has been standardized to ensure the reliable and cost-effective production of high quality reagents that constitute an essential support for both the research of the laboratory and the broader scientific community of NYUAD.